Proper classification of medical waste using the correct coduri deseuri medicale is no longer just an operational task—it is a critical part of a company's sustainability reporting under new EU regulations. These codes, defined by the European Waste Catalogue (EWC), form the data backbone for tracking waste streams and demonstrating responsible management for the Corporate Sustainability Reporting Directive (CSRD).
Your Guide to Medical Waste Codes and CSRD Compliance
Think of the system of coduri deseuri medicale as a direct line to regulatory compliance. Correctly applying these codes is not just a chore for healthcare facilities; it's a critical data point for any company’s environmental, social, and governance (ESG) disclosures, especially under the new European Sustainability Reporting Standards (ESRS).
Our team sees firsthand that accurate waste classification is the very first step in building an audit-proof data trail for your ESG reports. It turns what can feel like a compliance headache into a scalable process that you can manage effectively.
Proper coding is fundamental for a few key business reasons:
- Tracking Scope 3 Emissions: Waste disposal is a significant source of indirect greenhouse gas emissions. Clean data here leads to precise carbon footprint calculations.
- Demonstrating Circularity: Segregating waste correctly from the start helps identify opportunities for recycling and resource efficiency—a central theme in ESRS E5 (Resource use and circular economy).
- Ensuring Legal Compliance: Following national and EU waste laws helps avoid significant fines and protects your business reputation. For a comprehensive overview of equipment disposal, this Must Know Guide To Safe And Legal Disposal Of Medical Equipment is a useful resource.
The Link Between Waste Codes and ESG Reporting
The connection is straightforward: regulators, investors, and clients want to see transparent proof of your environmental impact. Your waste data provides tangible evidence, showing you manage your operations responsibly.
For example, the distinction between hazardous waste (marked with an asterisk, like 18 01 03*) and non-hazardous waste (like 18 01 04) has major consequences for both your disposal costs and your reported environmental footprint.
From our experience in ESG audits, we see regulators increasingly scrutinising waste data trails. A company that cannot show how it classifies, tracks, and disposes of its waste will struggle to produce a credible CSRD report.
To help your business get a handle on the classification logic, here is a clear overview of the main medical waste categories before we dive into the detailed codes.
Primary Medical Waste Categories and Their ESG Significance
This table outlines the major types of medical waste, their sources, and why correct classification is critical for compliance and reporting.
| Waste Category | Description and Common Examples | Reporting Implication for CSRD and ESRS |
|---|---|---|
| Infectious Waste | Waste contaminated with blood, body fluids, or other potentially infectious materials. Examples include used swabs, culture dishes, and gloves. | Crucial for ESRS E1 (Climate change) due to emissions from treatment (e.g., incineration) and for ESRS S1 (Own workforce) regarding health and safety. |
| Pathological Waste | Human tissues, organs, body parts, and contaminated animal carcasses. | Has significant ethical and environmental implications. Incorrect disposal can lead to severe reputational damage and regulatory penalties. |
| Sharps Waste | Items that can cause cuts or puncture wounds, such as needles, scalpels, and broken glass. | A major health and safety risk. Reporting on sharps management demonstrates strong governance and commitment to worker safety under ESRS S1. |
| Pharmaceutical Waste | Unused, expired, or contaminated medicines, drugs, and vaccines. | Improper disposal can contaminate water sources. This waste is key for reporting on pollution (ESRS E2) and resource use (ESRS E5). |
| Non-Hazardous General Waste | Waste that has not been in contact with infectious agents, chemicals, or radioactive materials. Examples: office paper, packaging, and food waste from administrative areas. | Segregating this stream is essential for improving recycling rates and demonstrating progress on circular economy goals under ESRS E5. |
Understanding these categories is the first step. Each is tied to specific EWC codes, and mastering that connection makes your ESG reporting accurate and defensible.
Understanding the European Framework for Waste Classification
To properly use coduri deseuri medicale (medical waste codes), it is essential to understand the European legal framework they come from. The system is built on the Waste Framework Directive (2008/98/EC), which establishes the rules for waste management across the EU, including the waste hierarchy principle.
This directive is implemented through the European Waste Catalogue (EWC), applied via the List of Wastes (LoW) Decision. This list is the single source of truth for classifying any type of waste in the European Union. For any business operating here, its use is mandatory.
The Six-Digit Coding System
The EWC uses a six-digit code to give every waste stream a precise identity. The structure is logical, helping you narrow down the right code for what you’re discarding.
Here’s how it breaks down:
- First two digits (Chapter): This points to the industry or activity that created the waste. For medical waste, the key chapter is 18, which covers waste from human and animal healthcare.
- Second two digits (Sub-chapter): This gets more specific, identifying the process within that industry. For example, 18 01 is for wastes from natal care, diagnosis, treatment, or disease prevention in humans.
- Final two digits (Specific Waste Stream): This is the exact definition of the waste. For instance, 18 01 01 refers specifically to sharps.
This full six-digit code is mandatory on all waste transfer notes and official records. It creates a clear, transparent data trail from generation to final disposal or recovery.
Hazardous vs Non-Hazardous Waste
A critical function of the LoW is distinguishing between hazardous and non-hazardous waste. Any stream considered hazardous is marked with an asterisk (). For example, infectious waste is coded as 18 01 03, while general, non-hazardous waste from medical care receives the code 18 01 04.
This simple asterisk has significant implications for your business. It dictates everything—segregation, packaging, labeling, transport, and disposal—which directly affects operational costs and legal risks. Misclassifying hazardous waste as non-hazardous can lead to serious penalties and environmental damage.
For any company in Romania, this EU system is transposed into national law, making it the required standard. Mastering these codes is the foundation for a compliant and self-sufficient waste management process. This is especially relevant given Romania's broader waste sector challenges. While total waste generation fluctuates, the municipal waste recycling rate is just 11.9%, far from the EU's 60% target for 2030. Meanwhile, incineration—a primary disposal route for infectious medical waste—is a small but growing part of the national strategy. More data on Romania's waste profile is available from the European Environment Agency.
How can a company be sure its waste data is accurate enough for an ESG audit? The answer starts with correctly applying these EWC codes at the point of waste generation.
This framework is not just another regulatory hurdle. It’s the data architecture that supports credible ESG reporting for regulations like the CSRD (Corporate Sustainability Reporting Directive) and platforms such as EcoVadis. It turns compliance into a structured, manageable activity.
Chapter 18 Medical Waste Codes Explained
This section is a practical reference. Getting your coduri deseuri medicale right is the foundation of a transparent waste stream—a non-negotiable for accurate carbon footprinting and defensible ESG reporting. From our team’s experience in ESG audits, this is where daily operations and compliance meet.
Chapter 18 of the European Waste Catalogue (EWC/LoW) is dedicated to waste from human or animal healthcare and related research. It is broken down into two main sub-chapters: 18 01 for waste from human care and 18 02 for animal care. Each entry provides a specific code, with hazardous streams clearly marked by an asterisk (*).
To help your teams classify waste correctly at the source, this simple decision tree shows how the initial sorting process should work.
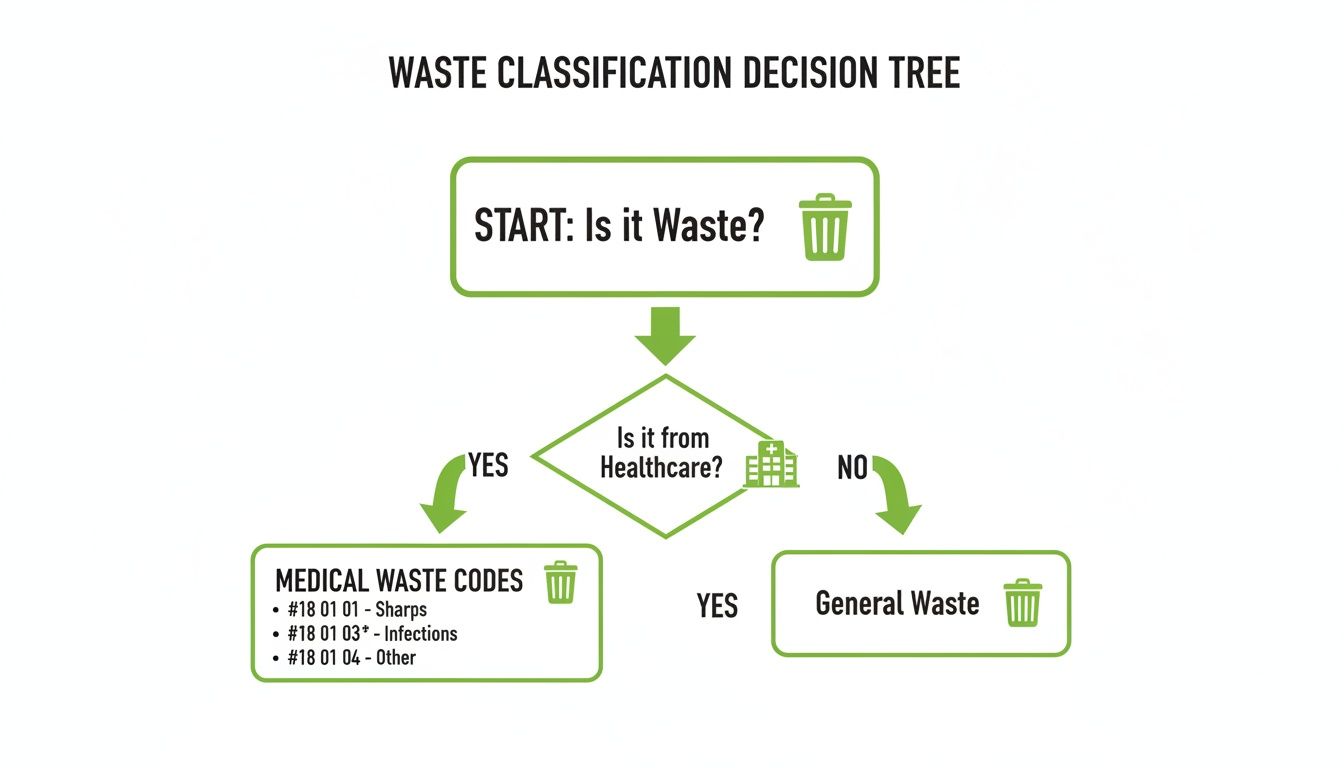
As the flowchart shows, the first question is whether the waste comes from a healthcare activity. This decision point triggers the need to apply the correct coduri deseuri medicale.
For a quick and practical reference, we’ve organized the most common codes into a single table. Below, you'll find more detailed explanations for each code.
Reference Table of Key Chapter 18 Medical Waste Codes
| Waste Code (EWC/LoW) | Official Description | Practical Examples | Is It Hazardous |
|---|---|---|---|
| 18 01 01 | Sharps (except for 18 01 03*) | Unused needles, uncontaminated scalpels, broken glass vials. | No |
| 18 01 02 | Body parts and organs including blood bags and blood preserves | Tissues, organs, anatomical parts from surgery. | No (but often treated as such) |
| 18 01 03* | Wastes whose collection and disposal is subject to special requirements in order to prevent infection | Used dressings, contaminated gloves, swabs, culture dishes, anything saturated with blood. | Yes |
| 18 01 04 | Wastes whose collection and disposal is not subject to special requirements in order to prevent infection | Uncontaminated gowns, packaging, disposable bedding, paper towels. | No |
| 18 01 06* | Chemicals consisting of or containing dangerous substances | Solvents, fixing solutions (e.g., from X-ray development), certain lab reagents. | Yes |
| 18 01 07 | Chemicals other than those mentioned in 18 01 06 | Non-hazardous cleaning agents, non-toxic lab reagents. | No |
| 18 01 08* | Cytotoxic and cytostatic medicines | Expired chemotherapy drugs, contaminated vials, IV bags and tubing used for administration. | Yes |
| 18 01 09 | Medicines other than those mentioned in 18 01 08 | Expired or unused non-chemotherapy pills, antibiotics, vaccines. | No (but disposal is regulated) |
| 18 01 10* | Amalgam waste from dental care | Old amalgam fillings, contaminated cotton rolls, chair-side traps containing mercury. | Yes |
This table serves as a quick lookup tool, but correct application requires understanding the nuances behind each code.
Codes for Human Healthcare Waste (18 01)
This sub-chapter covers all waste generated from natal care, diagnosis, treatment, or disease prevention in humans. Here’s a detailed breakdown of the codes your facility will encounter most often.
18 01 01 Sharps (except 18 01 03*)
This code is for non-infectious sharps, such as unused or uncontaminated needles, scalpels, or broken glass vials from a clean area. While not technically hazardous, the physical risk requires collection in rigid, puncture-proof containers.18 01 02 Body Parts and Organs
This refers to anatomical waste, including blood bags and blood preserves. Examples include tissues and organs removed during surgery. Although classified as non-hazardous, it is almost always treated as hazardous due to ethical considerations and local regulations, which typically mandate incineration.18 01 03* Wastes Whose Collection and Disposal Is Subject to Special Requirements
This is your main code for infectious waste. It covers anything contaminated with substances that could cause disease, including used dressings, swabs, contaminated gloves, and materials visibly saturated with blood. This waste is hazardous and must be packaged in designated yellow bags or containers for disposal via incineration or sterilisation.18 01 04 Wastes Whose Collection and Disposal Is Not Subject to Special Requirements
This code is for general, non-infectious medical waste, similar to municipal waste. It includes items like gowns, uncontaminated gloves, and packaging not in contact with infectious agents. It is not hazardous.
We see this all the time: misclassifying 18 01 04 waste as 18 01 03*. This common mistake unnecessarily inflates disposal costs and hazardous waste volumes, skewing ESG and CSRD data. Proper staff training on this distinction is crucial for cost control and accurate reporting.
Chemical and Pharmaceutical Waste Codes (18 01)
A significant portion of medical waste involves chemicals and pharmaceuticals, each with dedicated codes.
18 01 06* Chemicals Consisting of or Containing Dangerous Substances
This applies to hazardous chemicals used in medical or laboratory settings, like solvents or fixing solutions from X-ray development. This is hazardous waste.18 01 07 Chemicals Other Than Those Mentioned in 18 01 06
The counterpart for non-hazardous chemicals. It covers items like certain cleaning agents or non-toxic laboratory reagents. This is not hazardous.18 01 08* Cytotoxic and Cytostatic Medicines
This is for highly hazardous drugs, most of which are used in chemotherapy. It includes expired chemotherapy drugs and all materials used to administer them. This waste is highly hazardous and requires the strictest level of care.18 01 09 Medicines Other Than Those Mentioned in 18 01 08
This code is for all other non-cytotoxic pharmaceutical waste, such as expired non-chemotherapy pills, antibiotics, and vaccines. While not hazardous, its disposal is regulated to prevent environmental contamination.18 01 10* Amalgam Waste from Dental Care
Specifically for mercury-containing waste from dental fillings. Due to the presence of mercury, this is hazardous waste.
The codes for veterinary activities under sub-chapter 18 02 mirror this structure almost exactly. For example, 18 02 02* is the veterinary equivalent of 18 01 03*. Ensuring teams can distinguish between these human and animal streams is crucial for accurate record-keeping and building a reliable data inventory.
Practical Steps for Compliant Waste Management and Reporting
Let's translate regulations into on-the-ground execution. A compliant medical waste system is a chain of practical, connected steps that turns rules into a smooth workflow. From our experience, the foundation is the waste log. Get that right, and you have the reliable data needed to calculate a credible carbon footprint and demonstrate progress.
It starts at the source: the bin. Proper segregation is the single most important step. This is where your team decides if waste is hazardous or not—a decision that directly impacts safety, costs, and the integrity of your ESG data. For more detail, you can consult this complete guide to medical equipment disposal for hospitals and clinics.
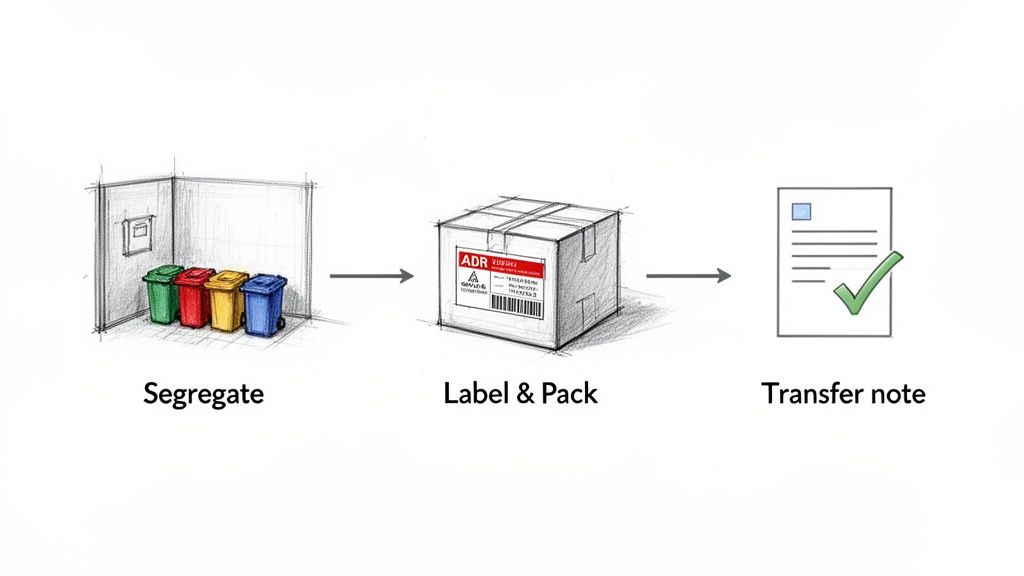
From Segregation to Record-Keeping
Once waste is sorted, the next step is correct labelling and packaging. Every container must be clearly marked with the right coduri deseuri medicale, the date, and the generating unit. For hazardous materials, specific packaging—often dictated by ADR transport regulations—is required to ensure safe handling.
The final operational step is creating an impeccable paper trail. Every time waste leaves your facility, it must be documented on a waste transfer note (in Romania, the formular de transport deșeuri). This document is the legal proof of handover to an authorised waste collector.
So, how can you be sure your waste data will stand up to an audit for a CSRD disclosure or an EcoVadis assessment? It all comes down to the integrity of these daily records. Digital, centralised logs are infinitely more reliable than paper files scattered across departments.
The COVID-19 pandemic threw this process into sharp relief. During Romania's state of emergency, medical waste generation grew to an estimated 4,312 tons, with 1,007.7 tons being infectious waste requiring incineration. For any company in healthcare, knowing the correct coduri deseuri medicale is now fundamental for CSRD compliance and Scope 3 emissions tracking. The poor data availability from that period was a wake-up call for the need for better internal inventories.
Building an Audit-Ready Workflow
To ensure your data is solid and defensible, focus on these key actions:
Standardise Procedures: Create simple, visual guides for waste segregation and post them where waste is generated. Use the official colour-coding system (yellow for infectious, black for non-hazardous).
Train Your People: Your staff are your first line of defence. We combine delivery with training so teams become autonomous. Regular, practical training ensures they understand the waste codes and the risks of misclassification.
Digitise Records: Replace paper logs. Use a simple spreadsheet or dedicated software to track waste types, quantities, collection dates, and destinations. This makes data retrieval for CSRD or EcoVadis reporting faster and more accurate.
Verify Your Partners: Ensure your waste transport and disposal partners are fully authorised and provide complete, accurate documentation for every pickup. These documents are a critical part of your own compliance records.
By embedding these steps into daily routines, waste management becomes a source of valuable data. This data doesn't just prove compliance; it provides insights to cut costs and shrink your carbon footprint. For a closer look at day-to-day requirements, see our guide on medical waste management.
Integrating Medical Waste Data into Your ESG Strategy
Your medical waste data is a strategic asset for any modern Environmental, Social, and Governance (ESG) plan, not just a compliance task. The records you keep on coduri deseuri medicale offer a direct look into your operational efficiency and environmental footprint.
It's time to put that data to work. By linking your waste volumes and disposal methods—like incineration versus landfill—to your carbon footprint, you can see their true impact on greenhouse gas emissions. This transforms mandatory record-keeping into a powerful tool for proactive management.
From Compliance Data to Strategic Insights
The information you gather for medical waste codes feeds directly into the most critical parts of your ESG reporting. It’s the tangible proof that strengthens your credibility with investors, customers, and partners.
The goal is to move from reactive reporting to active management, using this data to pinpoint reduction opportunities and build a more resilient, efficient business.
But how do you turn basic waste logs into a strategic advantage for your sustainability report? It comes down to connecting operational data to key ESG frameworks.
Here’s a practical look at how this data integration works:
Carbon Footprint (Scopes 1, 2, & 3): Waste disposal, particularly through incineration or transport, generates significant Scope 3 emissions. Accurate data on your waste volumes, correctly classified by coduri deseuri medicale, allows for precise and credible carbon footprint calculations.
Double Materiality Assessment: Your waste data is fundamental for a double materiality assessment under the Corporate Sustainability Reporting Directive (CSRD). It helps you identify both your operational impacts on the environment (like pollution) and the financial risks to your company (such as rising disposal costs).
ESRS Disclosures: This data is essential for reporting under specific European Sustainability Reporting Standards. It directly supports ESRS E1 (Climate change) by quantifying waste-related emissions and ESRS E5 (Resource use and circular economy) by showing how you manage resources and waste streams.
Strengthening Your Position with Stakeholders
Beyond regulatory demands, solid waste data management boosts your performance on third-party platforms that partners and clients use for evaluation.
Platforms like EcoVadis and CDP (formerly the Carbon Disclosure Project) include detailed questions on waste management policies and reduction initiatives. Providing clear, data-backed answers from your logs can significantly improve your scores. It proves your environmental management is robust, not just a set of claims.
We consistently see that companies with granular, well-organised waste data perform better in ESG assessments. It signals operational control and a serious commitment to environmental stewardship that goes beyond surface-level claims.
This organised approach allows you to answer stakeholder questions with confidence. It demonstrates you have systems in place not only to track your impact but also to actively manage it. In a market that values provable sustainability, this transparency builds trust and becomes a competitive differentiator.
Common Mistakes in Medical Waste Classification and How to Avoid Them
Getting waste classification wrong creates real business problems—legal penalties, environmental damage, and skewed sustainability reports. Drawing from our team’s experience with ESG audits and data verification, we see the same mistakes with coduri deseuri medicale happen repeatedly. These errors are costly but entirely avoidable.
Getting classification right from the start is about more than compliance. It’s about building a robust internal system, cutting operational risk, and ensuring your data can stand up to scrutiny. It's the foundation of a reliable waste management programme.

Over-Classifying Non-Hazardous Waste as Hazardous
This is the most frequent and expensive error we see. Out of caution, staff often throw non-infectious waste (Code 18 01 04) into yellow bags designated for infectious waste (Code 18 01 03*).
- The Business Risk: This practice inflates disposal costs, as treating hazardous waste is significantly more expensive. It also distorts your ESG data, painting a picture of higher risk and environmental impact than what is actually true.
- The Solution: Post clear, visual segregation guides where waste is generated. More importantly, run regular, hands-on training to give your team the confidence to differentiate between infectious and non-infectious materials.
Improper Segregation of Cytotoxic and Pharmaceutical Waste
Cytotoxic waste (Code 18 01 08*) is exceptionally hazardous and demands its own specific handling protocol. We sometimes see it mixed with general infectious waste. Similarly, non-hazardous expired medicines (18 01 09) can end up in general waste.
The legal and reputational fallout from mishandling cytotoxic drugs is severe. A single slip-up can trigger major fines and damage the trust of patients, regulators, and the public.
- The Business Risk: Incorrect sorting puts staff and the community at serious health risk. It’s a direct violation of hazardous materials regulations, leading to harsh penalties and reputational damage.
- The Solution: Use dedicated, clearly labelled purple containers exclusively for cytotoxic waste and set up a separate, secure collection process. For expired pharmaceuticals, establish a protocol for returning them to authorised handlers.
Failing to Code Mixed Waste Streams Correctly
What happens when different types of waste are thrown into the same container? The "precautionary principle" kicks in, meaning the entire mix must be classified, managed, and paid for under the most hazardous code applicable.
- The Business Risk: A small amount of infectious material contaminates a whole container of non-hazardous waste. This forces you to treat and pay for the entire volume as hazardous, undermining cost control. Solid record-keeping is crucial for tracking this. For more guidance, learn more about waste management records in our detailed article.
- The Solution: Emphasize segregation at the source. If some mixing is unavoidable, ensure staff are trained to apply the correct "worst-case" code (18 01 03* instead of 18 01 04) and document why it happened. This reinforces the need for better sorting practices.
Frequently Asked Questions About Medical Waste Codes
Working with coduri deseuri medicale always brings up practical questions for operations and compliance teams. It’s a common pain point. Here, we provide direct answers to the questions we hear most often during waste management and ESG reporting projects.
What Is the Difference Between Chapter 18 01 and 18 02 Codes?
Getting this right is fundamental to accurate record-keeping. Chapter 18 01 codes are strictly for waste generated from human healthcare—hospitals, clinics, dental practices, and outpatient centres.
In contrast, Chapter 18 02 codes cover waste from veterinary and animal research activities. This includes vet clinics, animal hospitals, and labs working with animals. Although the logic and waste types are similar (e.g., infectious waste, sharps), keeping these two streams separate is non-negotiable for regulatory compliance and accurate ESG reporting.
How Do I Classify a Mixed Medical Waste Stream?
This is a classic problem. If different types of medical waste end up in the same container and you cannot separate them, the "precautionary principle" applies. This rule means the entire container must be classified, handled, and recorded under the code for the most hazardous item inside.
For instance, if a bag contains both non-infectious dressings (code 18 01 04) and a few used, infectious sharps (code 18 01 03), the entire bag becomes hazardous infectious waste and must be documented as 18 01 03.
This principle is a vital risk-mitigation step, but it can drive up disposal costs and inflate your hazardous waste figures. It’s the single best argument for why good segregation at the source isn’t just a best practice—it’s a financial and compliance necessity.
Can I Use These Codes for Waste from a Cosmetics Clinic?
Yes, and you must. The use of Chapter 18 codes is based on the nature of the waste, not the type of facility. Any business generating waste with similar risks to medical waste must use the European Waste Catalogue for classification.
- Cosmetics clinics performing treatments that generate sharps, like needles for injections, must use the correct sharps code.
- Research laboratories creating chemical or biological waste should look to Chapter 18 or other relevant chapters (like Chapter 07 for chemical process wastes).
The key is to run a proper risk assessment on the waste itself. By looking at its properties—is it infectious, cytotoxic, or chemically hazardous?—you can pinpoint the right code and keep your organisation compliant.
At ECONOS 🌱, we specialise in turning complex regulatory demands like waste classification into clear, manageable processes. We don’t just deliver a service; we provide practical training that empowers your teams to build autonomous, audit-ready ESG systems.
Find out how we can support your CSRD and EcoVadis reporting at https://www.econos-esg.com.

.png)
